The search for effective and non-invasive methods to administer drugs and treat diseases has led to the development of innovative technologies. Among these technologies, electroporation has emerged as a revolutionary tool with amazing applications in various fields.
Electroporation is a technique that uses short, high-voltage electrical pulses to temporarily modify the permeability of cell membranes, allowing the entry of therapeutic molecules, active ingredients or genetic material into cells.
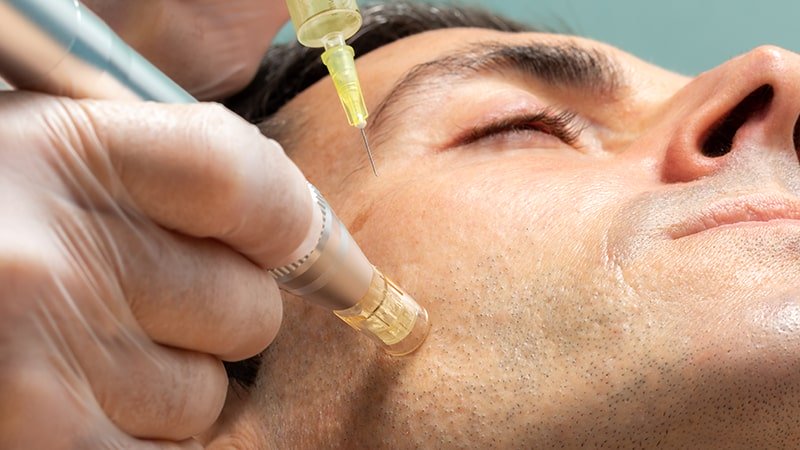
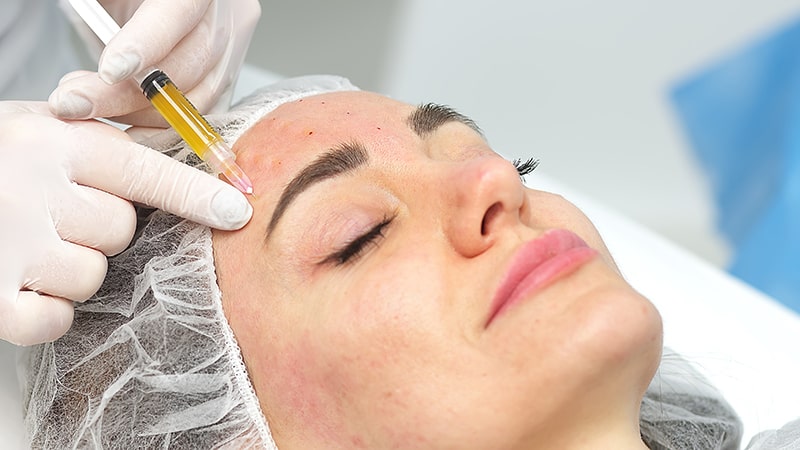
In the sector to which we belong, transdermal electroporation has been a revolution in aesthetic medicine, due to its efficacy, ease of use and rapid results.
However, this technique has also proven to be a versatile and effective tool for various applications in the field of medicine and biotechnology. Below, we review its most interesting applications.
Origin of electroporation
Although it seems to be a recent technology, the truth is that it was discovered in the 1970s by German scientists Pohl and Suessmilch. They observed that applying electrical pulses to cells caused changes in the structure of cell membranes, temporarily opening small pores in them, which allowed the passage of molecules that normally could not pass through the lipid barrier of the cell membrane.
The electroporation process is based on two main mechanisms: electrorepulsion and electroosmosis. During electrorepulsion, the electrical pulses induce a repulsive force that causes pores to form in the cell membrane. On the other hand, during electroosmosis, electrical pulses generate a flow of solvent through the pores, facilitating the transport of molecules into the cellular interior.
Uses of electroporation in medicine and biotechnology
Thanks to its ability to open new frontiers in disease research and treatment, electroporation is positioned as a promising technology with significant potential.
Drug administration
One of the great advances of electroporation is its ability to improve drug delivery. By applying electrical pulses, electroporation temporarily opens the cell membrane, allowing the entry of larger, electrically charged therapeutic molecules through the lipid barrier.
This technique has shown promising results in drug delivery, especially in the treatment of cancer, skin diseases and genetic diseases.
An example of this is in the treatment of skin cancer, specifically in the case of basal cell carcinoma and squamous cell carcinoma. These forms of skin cancer are often treated with topical therapy, and electroporation has proven to be an effective option to improve drug penetration and increase efficacy.
In this case, a gel or cream containing the anticancer drug is used. Electrodes are then applied to the affected area of the skin and short-duration, high-intensity electrical pulses are generated. These electrical pulses temporarily open the pores in the skin cells, allowing greater drug entry through the lipid barrier.
Electroporation significantly improves drug delivery to the skin tumor, which may increase efficacy and reduce systemic side effects associated with conventional treatments. In addition, it has been observed that this technique can increase the penetration of drugs into the deeper layers of the skin, which is especially relevant in more invasive tumor lesions.
Gene therapy
Before reaching the aesthetic sector, medical research observed how valuable electroporation could be for gene therapy, as a tool to treat hereditary or genetically acquired diseases.
By applying electrical pulses to target cells, nucleic acids, such as DNA or RNA, can be safely and efficiently introduced into cells. This opens up a world full of possibilities, since it allows the transfer of genetic material to the affected cells, i.e. it gives the ability to correct genetic mutations or activate specific genes for the treatment of diseases such as cancer, metabolic disorders and rare genetic diseases.
Duchenne muscular dystrophy is a degenerative and debilitating genetic disease caused by a mutation in the gene encoding the dystrophin protein. In patients with this disease, electroporation has been used as an approach to deliver a functional version of the dystrophin gene to the affected muscle cells.
In this case, muscle stem cells (also known as satellite cells) are isolated from the patient and a normal version of the dystrophin gene is introduced into them using a viral vector or a DNA plasmid. The cells are then subjected to electroporation, where electrical pulses are applied to allow the genetic material to enter the interior of the cells.
Once the cells have been genetically modified, they can be reintroduced into the patient by grafting or localized injection. These modified cells are expected to produce the functional dystrophin protein, which could improve muscle function and slow down the progression of the disease.
These techniques are still under development and research, but have already demonstrated great potential.
Drug development
In drug research and development, electroporation is used to introduce fluorescent molecules or genetic probes into living cells, allowing the monitoring and analysis of various biological processes in real time. In addition, electroporation is used in the generation of transgenic animal models and in the production of genetically modified animals to study the function of specific genes and to evaluate the efficacy of new drugs.
During the drug development phase, it is critical to understand the ability of a compound to enter target cells and exert its therapeutic action. Electroporation is used in cell permeability assays to assess the ability of a compound to cross the cell membrane and gain access to the cell interior.
In this case, cells are subjected to electroporation in the presence of the compound under evaluation. The electrical pulses allow the compound to enter the cells and then analyze and measure to determine the amount of compound that has entered the cells and evaluate its efficacy.
This assay technique is particularly useful when investigating compounds with low cell permeability, such as certain drugs with unfavorable physicochemical properties for cell entry.
Electroporation is also used in controlled drug delivery studies. In this case, drugs are encapsulated in liposomes or nanoparticles and electrical pulses are applied to release the drugs in a controlled manner at the desired site. In this way, therapeutic efficacy can be improved and side effects reduced by allowing a precise and localized release.
Tissue regeneration
By applying electrical pulses in combination with the administration of growth factors, wound healing, bone regeneration and tissue healing can be accelerated. This application has also been used in the treatment of sports injuries and rehabilitation after surgery, offering a non-invasive and efficient therapeutic option.
In the case of bone regeneration, low-intensity, short-duration electrical pulses are applied to the area of the bone defect. These pulses stimulate cells such as osteoblasts and osteocytes, promoting cell migration and proliferation, as well as extracellular matrix synthesis.
Electroporation facilitates the entry of bioactive molecules, such as growth factors and scaffold material, into bone cells, thereby enhancing bone tissue regeneration and repair. In addition, this technique can improve local vascularization and promote the formation of new blood vessels, which is crucial for nutrient supply and cell survival in the affected area.
In terms of tissue regeneration, the electrical pulses support the migration and proliferation of dermal and epidermal cells, promoting wound healing and the formation of functional skin tissue.